Case Series/Study
(CS-137) Transformable Powder Dressing Reduces Wound Volume in Chronic Refractory Wounds
Friday, April 28, 2023
7:15 PM - 8:30 PM East Coast USA Time
Introduction: The prevalence of chronic wounds continues to increase to epidemic proportions in the world and currently affects 6.7 million people in the United States. Chronic wounds remain challenging to heal and often do not respond to standard of care (SOC) treatments, necessitating innovative and novel therapies.
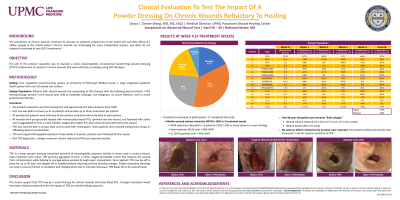
Methods: The objective was to determine the impact of a novel, biocompatible, non-occlusive transforming powder dressing (TPD) on wound healing trajectories in chronic wounds that were refractory to healing using SOC therapies. Five tertiary wound healing centers In a large academic center identified patients with chronic wounds that were not responding to SOC therapy. All selected patients were informed of the product evaluation, and agreed in the testing of a Class 1 medical device listed with the FDA as a “dressing, wound, hydrogel” that can be left on the wound for up to 30 days. All wounds were prospectively treated with methacrylate-based TPD, sprinkled into the wound, and hydrated with saline until it aggregated to form a moist, flexible, oxygen-permeable film that contoured and adhered to the wound. Patients were followed for a total of 3 weeks. The primary outcome measure was change in percent volume reduction (PVR) measured weekly for 3 weeks. Complications and intolerance to the TPD were recorded.
Results: Of the 19 initially enrolled patients, 17 patients completed the study. Wound subtypes included post- surgical (n=3), venous leg ulcers (n=8), diabetic foot ulcers (n=4), pressure injury (n=3), and trauma wounds (n=1). In the intent-to-treat group; 12 out of 19 wounds (63%) exceeded 50% PVR after 3 weeks, indicating a positive healing trajectory. There were no adverse events reported.
Discussion: TPD may be a useful therapy in chronic wounds that have failed standard of care, with a positive healing trajectory at 3 weeks. Extended clinical evaluation to determine the percentage of and time to complete healing with the addition of TPD is warranted.
Methods: The objective was to determine the impact of a novel, biocompatible, non-occlusive transforming powder dressing (TPD) on wound healing trajectories in chronic wounds that were refractory to healing using SOC therapies. Five tertiary wound healing centers In a large academic center identified patients with chronic wounds that were not responding to SOC therapy. All selected patients were informed of the product evaluation, and agreed in the testing of a Class 1 medical device listed with the FDA as a “dressing, wound, hydrogel” that can be left on the wound for up to 30 days. All wounds were prospectively treated with methacrylate-based TPD, sprinkled into the wound, and hydrated with saline until it aggregated to form a moist, flexible, oxygen-permeable film that contoured and adhered to the wound. Patients were followed for a total of 3 weeks. The primary outcome measure was change in percent volume reduction (PVR) measured weekly for 3 weeks. Complications and intolerance to the TPD were recorded.
Results: Of the 19 initially enrolled patients, 17 patients completed the study. Wound subtypes included post- surgical (n=3), venous leg ulcers (n=8), diabetic foot ulcers (n=4), pressure injury (n=3), and trauma wounds (n=1). In the intent-to-treat group; 12 out of 19 wounds (63%) exceeded 50% PVR after 3 weeks, indicating a positive healing trajectory. There were no adverse events reported.
Discussion: TPD may be a useful therapy in chronic wounds that have failed standard of care, with a positive healing trajectory at 3 weeks. Extended clinical evaluation to determine the percentage of and time to complete healing with the addition of TPD is warranted.

.png)